Ra’iisul Wasaare Xamsa oo si diirran loogu soo dhaweeyay Minneapolis Ra’iisul Wasaaraha Xukuumadda Jamhuuriyadda Federaalka Soomaaliya, Mudane...
Guddoomiye Sheekh Aadan Oo Soo Xiray Munaasabada Lagu Soo Bandhigaayay Kaarka Aqoonsiga Muwaadinka Guddoomiyaha Golaha Shacabka Baarlamaanka...
Madaxweynaha Jamhuuriyadda Federaalka Soomaaliya Mudane Xasan Sheekh Maxamuud oo maanta qaatay kaarka Aqoonsiga daweynaha ayaa ka hadlay...
Madaxweyne Xasan Sheekh “Waxaad Ku Jirtaan Dagaal Waafaqsan Mabaadii’da Shareecada Islaamka” Madaxweynaha Jamhuuriyadda Federaalka Soomaaliya Mudane Xasan...
Apple waxay ku qabatay xafladeeda furitaanka iPhone sanadlaha ah Talaadada Apple Park, oo ah xarunta shirkadda ee...
WAA QEYBTII 3AAD ogeyn, ayaa hoos u eegaysa inanteeda. Dareen ayay ka qaadatay, waxba se ma ay...
Guddoomiyaha Gobolka Banaadir Ahna Duqa Muqdisho Mudane Yuusuf Xuseen Jimcaale (Madaale) ayaa xarunta aqalka dowladda hoose ee...
Ra’iisul Wasaare Xamsa oo Daah-furay Mashaariic Caafimaad oo Dalka laga hirgelinayo Muqdisho, Sebtembar 11, 2023—Ra’iisul Wasaaraha Xukuumadda...
Dhulgariirkii ugu dhimashada badnaa ee ku dhuftay dalka Marooko muddo tobanaan sano ah ayaa waxaa ku dhintay...
Guddoomiyaha Gobolka Banaadir Ahna Duqa Muqdisho Mudane Yuusuf Xuseen Jimcaale (Madaale) ayaa xarunta aqalka dowladda hoose ee...
THIS IS PART 3 like a noodle, that it started to beat. Success without understanding what was...
WAA QEYBTII 3AAD baasto la moodo, in ay ku garaacdo bilowday. Najax oon fahmahayn waxa eeggan socda...
Ra’iisul Wasaare Ku Xigeenka Xukuumadda JFS, Mudane Saalax Axmed Jaamac oo khudbad ka jeediyay shirka Isbeddelka Cimillada...
Madaxweyne Xasan Sheekh Oo Kulan La Qaatay Hogaannada Taliska Ciidanka Xoogga Dalka Madaxweynaha Jamhuuriyadda Federaalka Soomaaliya Mudane...
Ubadka Madaxweynaha Iyo Furunta Dagaalka Madaxweyne Hassan Sheikh Mohamud Allaha dhaafee kaligii kuma aha furunta dagaalka ee...
THIS IS PART 2 – Bisinna, you didn’t see anything in the show. – Oh, who is...
WAA QEYBTII 2AAD – Bisinka, show waxba maad arkayn, way ka dhex baxdayba ka yar. – Alla...
𝗥𝗮’𝗶𝗶𝘀𝘂𝗹 𝗪𝗮𝘀𝗮𝗮𝗿𝗲 𝗫𝗮𝗺𝘀𝗮: “𝗟𝗮𝗺𝗮 𝗔𝗾𝗯𝗮𝗹𝗶 𝗗𝗼𝗼𝗻𝗼 𝗶𝗻 𝗟𝗮 𝗫𝗮𝗴𝗮𝗹 𝗗𝗮𝗮𝗰𝗶𝘆𝗼 𝗛𝗮𝗹𝗴𝗮𝗻𝗸𝗮 𝗗𝗮𝗹𝗸𝗮 𝗟𝗼𝗼𝗴𝗮 𝗫𝗼𝗿𝗲𝘆𝗻𝗮𝘆𝗼 𝗞𝗵𝗮𝘄𝗮𝗮𝗿𝗶𝗶𝗷𝘁𝗮.” Ra’iisul Wasaaraha...
Madaxweyne Xasan Sheekh Maxamuud oo kormeer ku tagay xerada ciidamada Guutada Cali binu Abiidaalib ee degmada Maxaas...
Ra’iisul Wasaare Xamsa iyo Wafdi Uu Hoggaaminayo oo u Ambabaxay Dalka Qatar Ra’iisul Wasaaraha Xukuumadda Jamhuuriyadda Federaalka...
6 years goes to twenty, beauty goes to it In the mind, a warm light follows the...
6 sanno ayaa u raaca labaatanka, qurux ayaa u raacda maskaxda, laydh diirran ayaa u raacda qorraxda....
Madaxweynaha Galmudug mudane Axmed Cabdi Kaariye ayaa gaaray deegaanno hoostaga degmada Ceelbuur oo dhawaan laga xoreeyay Khawaarijta...
Waraysiga uu Madaxweynihii hore ee Hirshabelle Maxamed Cabdi Waare siiyay Hanoolaato, waa mid laga dheehan karo quus...
Madaxweynaha Jamhuuriyadda Federaalka Soomaaliya Mudane Xasan Sheekh Maxamuud oo ka duulaya dardar galinta howgalada ciribtirka Khawaarijta ayaa...
Madaxweyne Xasan Sheekh Oo Si Kumeel-Gaarka Ah Ugu Shaqeynaya Magaalada Dhuusamareeb Ee Xarunta Dowlad-Goboleedka Galmudug Madaxweynaha Jamhuuriyadda...
A prominent cryptocurrency whale, renowned for its profitable trading exploits with Pepe (PEPE) tokens on Binance, encountered...
In a groundbreaking development, the Ethereum network is on the verge of implementing a radical update known...
TALISKA CIIDANKA XOOGGA DALKA SOOMAALIYEED OO FULINAYA HESHIISKII LA WAREEGISTA AMNIGA GUUD EE DALKA AYAA LA WAREEGAY...
MADAXWEYNE XASAN SHEEKH OO KULAN LA QAATAY WASIIRKA GAASHAANDHIGGA MARAYKANKA Madaxweynaha Soomaaliya ayaa kulan miro-dhal ah kula...
Guddoomiyaha Gobolka Banaadir Ahna Duqa Muqdisho Mudane Yuusuf Xuseen Jimcaale (Madaale) ayaa dhagax dhigay dib u casriyeenta...
President Hassan Sheikh Mohamud Strengthens Alliances, Celebrates Culture, and Promotes Peace and Prosperity The President of the...
Somalia People’s Assembly Engages in Productive Discussions on Legislative Agenda The Chairman of the Parliament, Mr. Sheikh...
Somalia has been a country rife with conflict and instability for decades. The impact of war and...
Arday is a new Somali television series that has garnered a lot of attention and praise since...
The 63rd anniversary of the foundation of the National Armed Forces is a day marked by the...
President of Somalia received the Secretary General of the United Nations at the Presidential Palace


President of Somalia received the Secretary General of the United Nations at the Presidential Palace
The President of Somalia received the Secretary General of the United Nations at the Presidential Palace The...
The Prime Minister of the Federal Government of Somalia, Mr. Hamse Abdi Barre, inaugurated the modern Jubba...
Here is the translation of the article you provided me with: The Somaliland opposition has recently spoken...
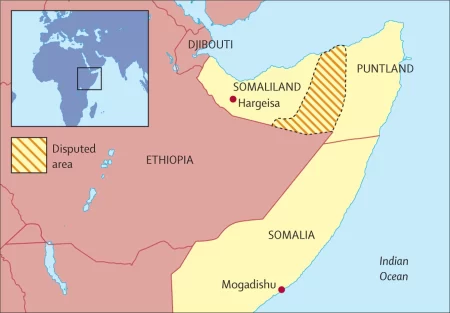
The government of Somaliland has issued a serious warning to the Puntland administration following a fierce battle...
The city of Mogadishu, the capital of Somalia, has declared a war on drugs and drug dealers....
Dissenting politicians are expelled by Somaliland’s Kulmiye Party. President Muse Bihi’s Kulmiye party, which is now in...
Multiple sources have confirmed that Abdirahman Duale Baileh, the former head of Somalia’s Treasury, has returned to...
Somaliland, a self-declared independent country in the Horn of Africa, is facing a new wave of violence...
Somaliland, a self-declared independent country in the Horn of Africa, is facing a new wave of violence...
The past few months have been marked by unusual and extreme weather events across the globe, causing...
Xaflad casho sharaf maqaamkeedu sarreeyo oo ay soo qaban qaabisay safaarada soomaaliya ee dalka ingiriiska ayaa xalay...
Mudo 32 sano kadib dowlada faderaalka soomaaliya waxa u suuragashay in ay saafarad ka furato dalka ingiriiska...
Wakaallada wararka Raashiya NOVOSTI ayaa sheegtay in diyaarad Helikobtar oo ay leedahay shirkadda UTair Corporation Helicopter lagu...
Ruushka ayaa si adag u cambaareeyay weerarkii foosha xumaa ee lagu gubay Qur’aanka Kariimka oo ka dhacay...